Abstract
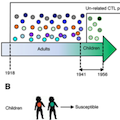
Novel influenza viruses often cause differential infection patterns across different age groups, defined as heterogeneous demographic susceptibility. This occurred during the A/H2N2 pandemic, when children experienced higher influenza attack rates than adults. As the recognition of conserved epitopes across influenza subtypes by CD8+ cytotoxic T lymphocytes (CTLs) limit influenza disease, we hypothesised that conservation of CTL antigenic peptides (Ag-p) in viruses circulating before the pH2N2-1957 may have resulted in differential CTL immunity. We compared viruses isolated in the years preceding the pandemic (1941–1957) to which children and adults were exposed, with viruses circulating decades earlier (1918–1940), which could infect adults only. Consistent with phylogenetic models, influenza viruses circulating 1941-1957, which infected children, shared with pH2N2 the majority (∼89%) of CTL-peptides within the most immunogenic nucleoprotein, matrix-1 and polymerase basic-1, thus providing evidence for minimal pH2N2 CTL escape in children. Our study, however, identified potential CTL immune evasion from pH2N2 irrespective of age, within HLA-A03:01+ individuals for PB1471-L473V/N476I variants and HLA-B15:01+ population for NP404-414-V408I mutant. Further experiments using the murine model of B-cell-deficient mice showed that multiple influenza infections resulted in superior protection from influenza-induced morbidity, coinciding with accumulation of tissue resident memory CD8+ T-cells in the lung. Our study suggests that protection against H2N2-1957 pandemic influenza was most likely linked to the number of influenza virus infections prior to the pandemic challenge rather than differential pre-existing CTL immunity. Thus, the regimen of a CTL-based vaccine/vaccine-component may benefit from periodic boosting to achieve fully protective, asymptomatic influenza infection.